Basics about Discharging
Learn how certain discharge loads will shorten battery life.
The purpose of a battery is to store energy and release it at a desired time. This section examines discharging under different C-rates and evaluates the depth of discharge to which a battery can safely go. The document also observes different discharge signatures and explores battery life under diverse loading patterns.
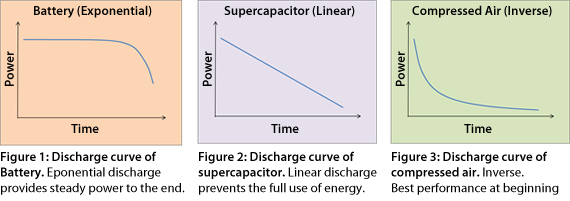
The electrochemical battery has the advantage over other energy storage devices in that the energy stays high during most of the charge and then drops rapidly as the charge depletes. The supercapacitor has a linear discharge, and compressed air and a flywheel storage device is the inverse of the battery by delivering the highest power at the beginning. Figures 1, 2 and 3 illustrate the simulated discharge characteristics of stored energy.
Most rechargeable batteries can be overloaded briefly, but this must be kept short. Battery longevity is directly related to the level and duration of the stress inflicted, which includes charge, discharge and temperature.
Remote control (RC) hobbyists are a special breed of battery users who stretch tolerance of “frail” high-performance batteries to the maximum by discharging them at a C-rate of 30C, 30 times the rated capacity. As thrilling as an RC helicopter, race car and fast boat can be; the life expectancy of the packs will be short. RC buffs are well aware of the compromise and are willing to both pay the price and to encounter added safety risks.
To get maximum energy per weight, drone manufacturers gravitate to cells with a high capacity and choose the Energy Cell. This is in contrast to industries requiring heavy loads and long service life. These applications go for the more robust Power Cell at a reduced capacity.
Depth of Discharge
Lead acid discharges to 1.75V/cell; nickel-based system to 1.0V/cell; and most Li-ion to 3.0V/cell. At this level, roughly 95 percent of the energy is spent, and the voltage would drop rapidly if the discharge were to continue. To protect the battery from over-discharging, most devices prevent operation beyond the specified end-of-discharge voltage.
When removing the load after discharge, the voltage of a healthy battery gradually recovers and rises towards the nominal voltage. Differences in the affinity of metals in the electrodes produce this voltage potential even when the battery is empty. A parasitic load or high self-discharge prevents voltage recovery.
A high load current, as would be the case when drilling through concrete with a power tool, lowers the battery voltage and the end-of-discharge voltage threshold is often set lower to prevent premature cutoff. The cutoff voltage should also be lowered when discharging at very cold temperatures, as the battery voltage drops and the internal battery resistance rises. Table 4 shows typical end-of-discharge voltages of various battery chemistries.
| End-of-discharge
Nominal |
Li-manganese 3.60V/cell |
Li-phosphate 3.20V/cell |
Lead acid 2.00V/cell |
NiCd/NiMH 1.20V/cell |
| Normal load
Heavy load or |
3.0–3.3V/cell 2.70V/cell |
2.70V/cell 2.45V/cell |
1.75V/cell 1.40V/cell |
1.00V/cell 0.90V/cell |
Table 4: Nominal and recommended end-of-discharge voltages under normal and heavy load.
The lower end-of-discharge voltage on a high load compensates for the greater losses.
Over-charging a lead acid battery can produce hydrogen sulfide, a colorless, poisonous and flammable gas that smells like rotten eggs. Hydrogen sulfide also occurs during the breakdown of organic matter in swamps and sewers and is present in volcanic gases and natural gas. The gas is heavier than air and accumulates at the bottom of poorly ventilated spaces. Strong at first, the sense of smell deadens with time, and the victims are unaware of the presence of the gas. (See BU-703: Health Concerns with Batteries.)
What Constitutes a Discharge Cycle?
A discharge/charge cycle is commonly understood as the full discharge of a charged battery with subsequent recharge, but this is not always the case. Batteries are seldom fully discharged, and manufacturers often use the 80 percent depth-of-discharge (DoD) formula to rate a battery. This means that only 80 percent of the available energy is delivered and 20 percent remains in reserve. Cycling a battery at less than full discharge increases service life, and manufacturers argue that this is closer to a field representation than a full cycle because batteries are commonly recharged with some spare capacity left.
There is no standard definition as to what constitutes a discharge cycle. Some cycle counters add a full count when a battery is charged. A smart battery may require a 15 percent discharge after charge to qualify for a discharge cycle; anything less is not counted as a cycle. A battery in a satellite has a typical DoD of 30–40 percent before the batteries are recharged during the satellite day. A new EV battery may only charge to 80 percent and discharge to 30 percent. This bandwidth gradually widens as the battery fades to provide identical driving distances. Avoiding full charges and discharges reduces battery stress. (See also BU-1003: Electric Vehicle.)
A hybrid car only uses a fraction of the capacity during acceleration before the battery is recharged. Cranking the motor of a vehicle draws less than 5 percent energy from the starter battery, and this is also called a cycle in the automotive industry. Reference to cycle count must be done in context with the respective duty.
Reference to discharge cycle or cycle count does not relate equally well to all battery applications. One example where counting discharge cycles does not reflect state-of-life accurately is in a storage device (ESS). These batteries supplement renewable energies from wind power and photovoltaic by delivering short-term energy when needed and storing if in excess. The time duration between charge and discharged can be in milliseconds; a typical battery state-of-charge is 40–60%. Rather than cycle count, coulomb counting may be used as a means of measuring wear and tear.