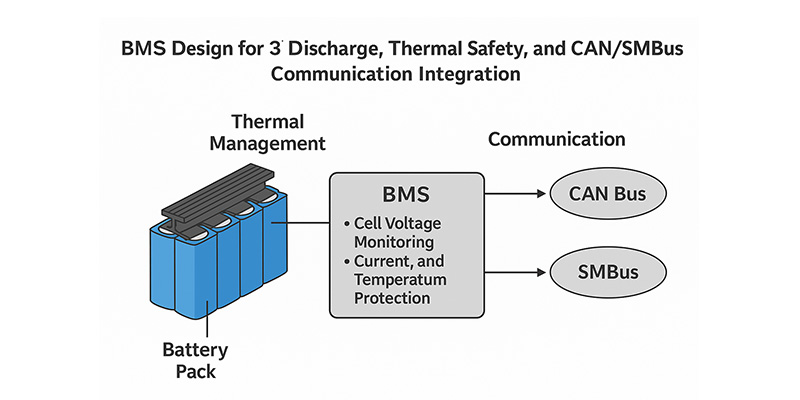
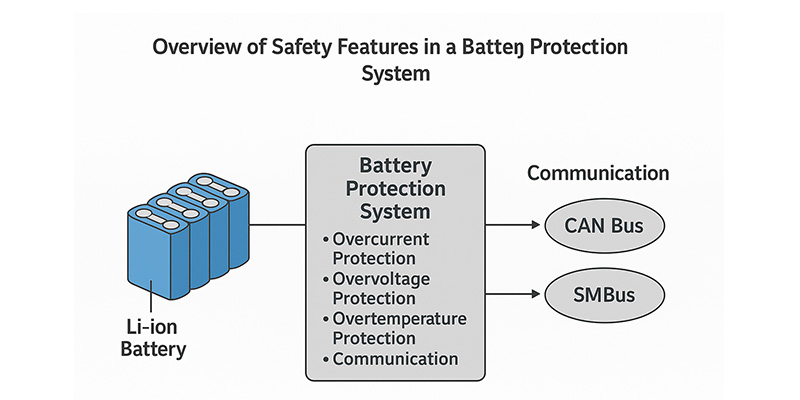
When it comes to high-performance lithium battery packs, especially those powering compact EVs, robots, and portable industrial equipment, safety and control are everything. At the heart of it all lies the Battery Management System (BMS).
A smart BMS not only protects the battery—it unlocks high discharge capabilities, ensures stable thermal performance, and allows seamless communication with host systems. In this article, we explore how advanced BMS design enables 3C continuous discharge, effective heat management, and dual communication support using CAN Bus and SMBus protocols—and how Himax has implemented these technologies in real-world custom battery solutions.
Why Advanced BMS Design Matters
Every lithium battery pack requires a BMS to manage charging, discharging, voltage balancing, and safety cut-offs. But in high-rate applications—such as e-bikes, delivery robots, or mobile power stations—the BMS becomes a critical performance enabler.
An ordinary BMS might limit your device’s power output or trigger premature shutdowns under high load. A well-engineered BMS, however, empowers your product with:
- Reliable 3C discharge rates(e.g., 30–45A continuous at 48V)
- Integrated thermal protectionfor extended safety
- Smart communicationwith host systems via CAN or SMBus
- Real-time diagnostics and error reporting
1. Enabling 3C Discharge Through Intelligent BMS Architecture
(keyword: 3c discharge, bms protection, high-rate battery pack)
A 3C discharge means the battery can safely output current equal to three times its capacity per hour. For example, a 10Ah pack with 3C capability can sustain a 30A discharge—ideal for motors, pumps, and actuators.
At Himax, we achieve this using:
- High-precision current sensors (shunt-based)
- Ultra-low resistance MOSFET switching arrays
- Custom-configured voltage and current thresholds
- Dynamic control logic to prevent overcurrent and short circuits
Case Example: Our 48V 44Ah battery pack delivers a steady 45A discharge (just above 1C) but is tested and certified for short bursts of up to 120A peak without triggering thermal cut-off.
This level of output is ideal for:
- Electric scooters and delivery bikes
- Mobile robots and logistics platforms
- High-power field tools and emergency equipment
2. Thermal Management: Staying Cool Under High Load
(keyword: battery thermal management, pcm cooling, heat control)
Discharging at high current naturally generates heat. Without thermal management, this heat can degrade the battery, damage components, or even cause failure.
At Himax, we design battery packs with:
- PCM (Phase Change Material)inserts that absorb and release heat passively
- Finned aluminum heat sinksto maximize air-cooled dissipation
- Optional forced-air or active liquid cooling systemsfor large industrial units
- Real-time thermal cutoffif any cell exceeds safe limits
Our compact 22.2V 28Ah pack for robotics, for example, uses a hybrid structure: PCM blocks embedded around the cells + passive air vents for lightweight, silent cooling.
With this approach, we maintain safe operation even at discharge currents above 3C in closed, compact housings.
3. CAN Bus vs. SMBus: Smart Communication with Host Systems
(keyword: bms can bus, smbus battery communication, smart battery protocol)
Communication between the battery and the rest of your system is just as important as raw performance. Himax BMS supports both CAN Bus and SMBus, depending on your application:
| Protocol | Best For | Features |
| CAN Bus | EVs, robotics, industrial equipment | High-speed (1 Mbps), robust, multi-node |
| SMBus | Portable devices, smart tools | Lightweight, I²C-based, real-time alerts |
Our dual-protocol BMS allows:
- Real-time SOC/SOH reporting(State of Charge / Health)
- Temperature and voltage broadcastto controllers
- Fault flagging and event history logging
- Firmware upgrades over the bus
If you’re developing a smart EV or a connected tool, this enables full system integration and battery analytics.
4. Himax’s OEM Approach to Integrated BMS Solutions
(keyword: custom battery bms, oem battery manufacturer)
Unlike off-the-shelf solutions, Himax provides end-to-end design for battery packs with tailored BMS configurations. Here’s what makes our offering unique:
✅ Custom BMS firmware – Set your thresholds, alerts, and logic
✅ Flexible communication setup – CAN, SMBus, UART, RS485 all available
✅ Heat optimization by design – Enclosure, cell layout, airflow modeling
✅ Compliance ready – UN38.3, IEC62619, CE, RoHS, UL standards
✅ Data logging + remote diagnostics – Optional flash memory support
From 22.2V 28Ah compact packs to 48V 60Ah industrial systems, we’ve helped dozens of OEMs worldwide build smarter, safer power systems.
5. Applications That Require Intelligent BMS Design
(keyword: high-discharge battery applications)
The demand for smarter, high-performance BMS is growing across:
- E-mobility: electric scooters, city bikes, cargo trikes
- Robotics: autonomous floor cleaners, patrol robots, warehouse AGVs
- Field Tools: power drills, cable splicers, rescue gear
- Medical & Military: mobile ventilators, communications gear
- IoT & Energy: hybrid solar kits, portable UPS systems
In all of these, 3C discharge, precise heat control, and secure communication are not optional—they’re mission-critical.
FAQ – Battery Management System for High-Reliability Packs
Q1: What is 3C discharge in a battery pack?
A: It means the pack can safely deliver current 3x its capacity per hour—e.g., 30A for a 10Ah pack.
Q2: Why is thermal protection important in high-rate batteries?
A: It prevents cell degradation, swelling, and fire risk—especially under constant high current.
Q3: What’s the benefit of using CAN or SMBus in a BMS?
A: These allow real-time battery monitoring, system coordination, and safer shutdown in case of faults.
Q4: Can Himax combine both CAN and SMBus in one pack?
A: Yes. We offer dual-protocol BMS for complex systems needing multi-layer control.
Q5: How customizable is Himax’s BMS solution?
A: Fully. We customize thresholds, connectors, cell configurations, firmware, and even casing.
Build Safer, Smarter Battery Packs with Himax
Whether you’re developing a high-powered electric vehicle or a precision robotic platform, battery safety and intelligence are non-negotiable. Himax’s custom BMS solutions bring together the best of protection, performance, and integration—designed to fit your product, not the other way around.
Contact us now to explore a custom battery pack with 3C discharge capability, PCM thermal control, and seamless CAN/SMBus communication. Let’s engineer your power advantage—together.