Understanding the Importance of Customized Lithium-Ion Batteries
Lithium-ion batteries are renowned for their high energy density, lightweight nature, and long lifespan. These attributes make them ideal for a multitude of applications, including:
-
Consumer Electronics: Smartphones, laptops, and wearable devices.
-
Electric Vehicles (EVs): Cars, bikes, and scooters.
-
Energy Storage Systems: Grid storage and backup power solutions.
-
Industrial Equipment: Power tools, medical devices, and robotics.
Given the diverse applications, the performance requirements for batteries can vary significantly. Off-the-shelf batteries might not always align with specific operational demands, leading to suboptimal performance or even safety concerns. This is where customized lithium-ion battery solutions come into play, offering configurations tailored to unique business requirements.

Identifying Your Business Requirements: The First Step in Customization
Before diving into battery specifications, it’s crucial to thoroughly assess your business needs. This foundational step ensures that the customized solution aligns perfectly with your operational goals.
Industry-Specific Demands
Different industries have distinct energy requirements. For instance:
-
Consumer Electronics: Prioritize compact size and high energy density to ensure longer device usage between charges.
-
Electric Vehicles: Demand batteries with high power output and rapid charging capabilities to enhance vehicle performance and reduce downtime.
-
Energy Storage Systems: Require batteries with long cycle life and excellent safety profiles to ensure reliable and sustainable energy supply.
Understanding these nuances helps in selecting a battery configuration that meets industry-specific challenges.
Performance Specifications
Key performance metrics to consider include:
-
Voltage: Determines the electrical potential difference. Ensure compatibility with your device’s operating voltage.
-
Capacity (mAh or Ah): Indicates the amount of charge the battery can hold. Higher capacity translates to longer operational time.
-
Discharge Rate (C-rate): Reflects how quickly a battery can be discharged. High-drain applications require batteries with higher discharge rates.
Accurately defining these parameters ensures that the battery performs optimally under expected operating conditions.
Environmental Considerations
Operating environments can significantly impact battery performance and longevity. Factors to evaluate are:
-
Temperature Range: Extreme temperatures can affect battery efficiency and safety. For instance, applications in cold climates might require batteries with low-temperature performance enhancements.
-
Ingress Protection: For environments exposed to dust or moisture, batteries should have appropriate IP ratings to prevent ingress and potential damage.
By accounting for environmental factors, you can enhance the reliability and lifespan of the battery in its intended application.
Selecting the Appropriate Battery Chemistry: Balancing Performance and Cost
The internal chemistry of a lithium-ion battery dictates its characteristics. Common chemistries include:
-
Lithium Iron Phosphate (LiFePO4 or LFP): Known for safety and long cycle life, making them suitable for energy storage and electric vehicles.
-
Nickel Manganese Cobalt (NMC): Offers a balance between energy density and safety, commonly used in power tools and electric vehicles.
-
Lithium Cobalt Oxide (LCO): Provides high energy density, ideal for consumer electronics but with a shorter lifespan compared to other chemistries.
Each chemistry presents a trade-off between performance attributes and cost. For example, while LFP batteries offer enhanced safety and longevity, they might have a lower energy density compared to NMC batteries. Evaluating these trade-offs in the context of your specific application is essential for optimal performance and cost-effectiveness.
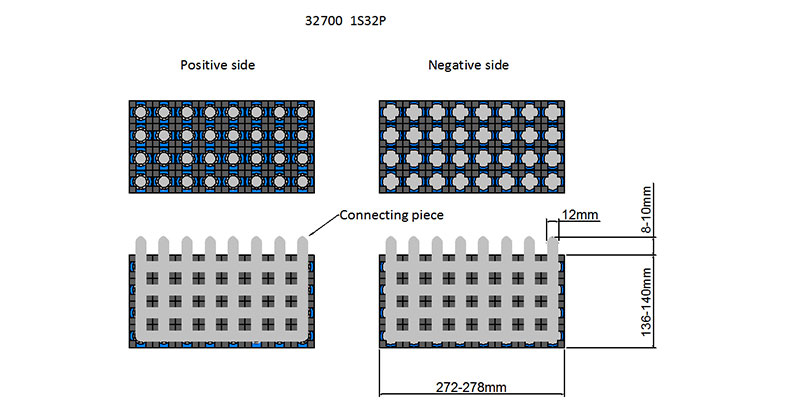
Designing Battery Shape and Size: Adapting to Spatial Constraints
The physical dimensions of the battery must align with the design constraints of the device or system it powers. Common battery formats include:
-
Cylindrical Cells: Robust and easy to manufacture, often used in power tools and electric vehicles.
-
Prismatic Cells: Rectangular-shaped, allowing for efficient use of space, commonly found in consumer electronics.
-
Pouch Cells: Flexible packaging that can be molded into various shapes, ideal for applications where space is at a premium.
Collaborating with battery manufacturers to design cells that fit seamlessly into your product can lead to better space utilization and, in some cases, improved performance.
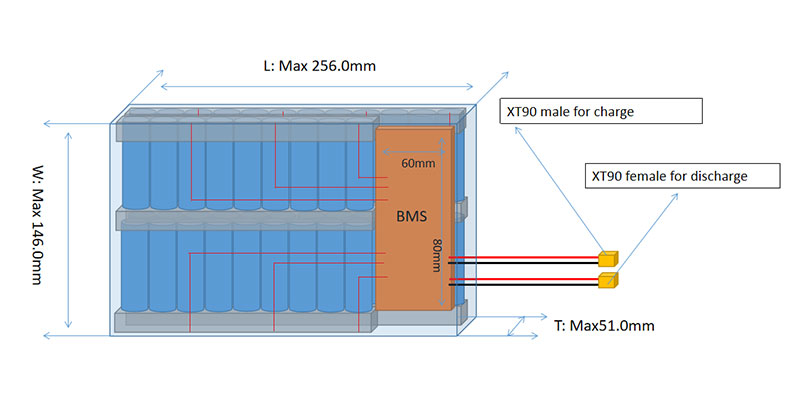
Implementing Battery Management Systems (BMS): Ensuring Safety and Efficiency
A Battery Management System (BMS) is pivotal in monitoring and managing the performance of lithium-ion batteries. Key functions of a BMS include:
-
Overcharge and Over-discharge Protection: Prevents the battery from operating outside its safe voltage range.
-
Temperature Monitoring: Ensures the battery operates within safe temperature limits, preventing overheating.
-
Cell Balancing: Maintains equal charge levels across all cells to optimize performance and extend lifespan.
For customized solutions, the BMS can be tailored to include features specific to the application’s needs, such as communication interfaces or specialized monitoring protocols.
Balancing Cost and Delivery Timelines: Managing Budget and Time Constraints
Customization often involves additional costs and extended lead times due to:
-
Design and Development: Engineering resources are required to design and test the custom battery.
-
Material Selection: Sourcing specific materials that meet the desired specifications.
-
Certification and Testing: Ensuring the battery meets industry standards and regulations.
Engaging with experienced manufacturers can help streamline the development process, potentially reducing costs and delivery times. It’s essential to communicate your budget and timeline constraints clearly to align expectations and project plans.
Ensuring Compliance and Certification: Meeting Regulatory Standards
Adherence to industry standards and obtaining necessary certifications is non-negotiable, especially for products entering regulated markets. Common certifications include:
-
UN38.3: Ensures the battery can withstand transportation conditions without posing hazards.
-
CE Marking: Indicates conformity with health, safety, and environmental protection standards for products sold within the European Economic Area.
-
UL Certification: Demonstrates that the product has been tested for safety risks.
Partnering with manufacturers familiar with these certifications can facilitate smoother market entry and reduce the risk of non-compliance.
Case Studies: Successful Implementation of Customized Lithium-Ion Batteries
Examining real-world applications can provide valuable insights into the customization process.
Case Study 1: Consumer Electronics
Challenge: A wearable technology company requires a lightweight battery with high energy density to ensure extended usage without compromising device comfort.
Solution: By opting for a prismatic LCO battery, they achieved the desired energy capacity within a compact form factor, enhancing user experience without adding bulk.
Case Study 2: Electric Vehicles
Challenge: An electric scooter manufacturer needed a battery capable of delivering high power output for rapid acceleration, with a long cycle life to ensure durability.
Solution: Implementing an NMC battery with a robust BMS provided the necessary power and longevity while also ensuring safety during high-discharge scenarios.
Case Study 3: Energy Storage Systems
Challenge: A renewable energy company sought a safe and cost-effective solution for storing solar energy in residential settings.
Solution: Utilizing LiFePO4 batteries, known for their long cycle life and thermal stability, the company provided homeowners with a reliable and sustainable energy storage solution. The integration of a smart BMS ensured efficient energy management and enhanced safety.
Partnering with the Right Manufacturer: Key Considerations
Selecting the right manufacturer for your customized lithium-ion battery is crucial for ensuring quality, performance, and compliance. Here are essential factors to consider:
1. Technical Expertise and Experience
Choose a manufacturer with extensive experience in lithium-ion battery design and production. Their expertise should align with your industry-specific requirements.
2. Customization Capabilities
Evaluate the manufacturer’s ability to tailor battery configurations, including chemistry, form factor, voltage, and BMS features, to your exact needs.
3. Quality Assurance and Testing
Ensure the manufacturer follows stringent quality control processes and conducts rigorous testing to meet industry standards, such as UN38.3, CE, and UL certifications.
4. Supply Chain and Scalability
A reliable manufacturer should have a well-established supply chain to ensure consistent material sourcing and the ability to scale production as your business grows.
5. Post-Sales Support and Warranty
Look for manufacturers offering comprehensive after-sales support, including troubleshooting, warranty coverage, and technical assistance.
Conclusion: Choosing the Right Customized Lithium-Ion Battery Solution
Selecting the right customized lithium-ion battery solutions requires careful evaluation of business needs, performance requirements, budget, and compliance factors. Businesses can ensure optimal performance and long-term reliability by considering key aspects such as battery chemistry, design, BMS integration, and industry certifications. Partnering with an experienced manufacturer is essential for achieving a tailored solution that meets specific application demands.
HIMAX Electronics is a leading provider of high-quality, custom lithium-ion battery solutions, offering advanced technology, rigorous safety standards, and tailored designs for diverse industries worldwide. Contact HIMAX Electronics today to explore how their expertise can power your business.